Graphene-based nanomaterials
Graphene is a single layer of carbon where atoms are packed by covalent sp2 bonds in a hexagonal honeycomb lattice [9]. The ultra-thin thickness and 2D morphological features give it an ultra-high specific surface area and unique physicochemical properties. As an allotrope of carbon, graphene possesses appropriate biocompatibility, excellent in-plane stiffness, exceptional antibacterial characteristics, and a sizable open surface capable of non-covalent interaction with biomolecules (Fig. 1A) [63,64,65]. Graphene is the first and most well-known 2D nanomaterial throughout the world. Various functionalized forms of graphene-based nanomaterials (GBN) have been developed and thoroughly investigated, mainly including graphene, graphene oxide (GO) and reduced graphene oxide (rGO) [66].
As an oxidized form of graphene, GO binds to oxygen-containing functional groups overhanging above or below the plane of nanomaterials through sp3 bonds, including carboxyl and carbonyl groups at the margins, and hydroxyl and epoxy groups on the basal surface [67]. This chemical modification allows GO more uneven than graphene, providing more active sites for covalent, electrostatic and hydrogen bond interactions with biomolecules. At the same time, the presence of oxygen also makes GO more hydrophilic than graphene. rGO is a structural intermediate of graphene and GO that can be readily obtained by thermal, chemical or ultraviolet photoreduction of GO [68]. In comparison to GO, rGO contains a lower oxygen concentration, surface charge and hydrophilic functional groups, appearing more hydrophobic [69].
Bone tissue engineering (BTE) has great potential in treating significant bone deficiencies resulting from severe trauma and nonunion of fractures. Conventional materials, like metals, ceramics and polymers, have been utilized extensively as BTE scaffolds. However, their biocompatibility and osseointegration are inadequate, and it is challenging to strike a balance between mechanical strength and biodegradation [70]. With the rapid development of 2D nanomaterials, remarkable biocompatibility, low toxicity and ultra-high specific surface area result in GBN an ideal biological scaffold for the delivery of growth factors and osteogenic medications [71]. In addition, the rough surface of GBN and the unique non-covalent π-π packing interaction also favor the concentration of osteogenic media such as dexamethasone and β-glycerophosphate on the nanoplatform, thereby mediating stem cell adhesion, diffusion, proliferation, cell signaling and osteogenic differentiation [72]. Research has demonstrated that mesenchymal stem cells (MSCs) derived from diverse origins exhibit comparable osteogenic tendencies on the surface of hypoxic graphene, and notable activation of angiogenesis factors and osteogenic transcription genes can be observed [73]. The carbon arrangement of graphene closely mimics the microenvironment of the organic bone extracellular matrix (ECM), enabling stem cells to adhere, proliferate, and differentiate under appropriate cues. Cell adhesion is the initiating event in this process; without it, cells have limited opportunities to produce ECM and receive differentiation signals. Owing to graphene’s planar structure, cells can adhere more closely to its surface, thereby facilitating differentiation into the osteogenic lineage. Experimental results confirm that graphene-based nanomaterials (GBN) upregulate the expression of multiple genes related to adhesion and ECM production in mesenchymal stem cells (MSCs) from various sources, thereby promoting osteoblast adhesion and triggering differentiation signals. As a result, MSCs from various sources exhibit similar osteogenic tendencies when cultured on graphene surfaces. Liu et al. [74] successfully synthesized nanographene oxide via employing electrochemical derivatization, which enhanced the formation of endothelial tip cells by recruiting endogenous lysophosphatidic acid and indicated efficient angiogenesis in in vitro and in vivo experiments.
It is crucial to remember that GBN exerts a wide range of complex impacts on stem cell development. The various forms and characteristics create a very different extracellular environment, thereby regulating stem cell behavior [75, 76]. When graphene is employed as a biological scaffold for culturing human mesenchymal stem cells (hMSCs), it demonstrates a strong ability to promote osteogenic differentiation while markedly inhibiting adipogenic differentiation. This phenomenon can be attributed to the denaturation of insulin—a key regulator of fatty acid synthesis—upon interaction with graphene, which results from strong π-π stacking interactions. In contrast, when hMSCs are cultured on hydrophilic graphene oxide (GO), their potential for adipogenic differentiation is enhanced [77]. This discrepancy may be due to the presence of oxygen-containing functional groups on GO, which disrupt the strong π-π bonding capabilities and the extensive sp2-conjugated structure inherent to graphene. Consequently, insulin adsorbed onto GO does not undergo denaturation. Furthermore, the high affinity between GO and insulin further promotes the adipogenic differentiation of hMSCs [78]. Collectively, these findings indicate that the distinct morphological and physicochemical properties of graphene-based nanomaterials (GBNs) play a pivotal role in directing the differentiation of hMSCs into specific tissue lineages.
Furthermore, graphene is frequently applied as a surface coating in combination with other traditional substances to improve degrading behaviors and enhance biological functions [79]. Zhou et al. [80] successfully loaded rGO on the surface of a graded porous hydroxyapatite (HA) scaffold. In vitro experiments showed that the degradation rate of the HA/rGO composite framework matched well with the rate of new bone formation and profoundly increased the proliferation and spontaneous osteogenic differentiation of bone marrow MSCs (BMSCs) (Fig. 1B). Likewise, Kang et al. [81] proved that introducing rGO to the surface of conventional titanium (Ti) scaffolds greatly reduced the surface roughness of the Ti matrix and boosted the osteogenic differentiation of BMSCs. As part of composite materials, GBN can strengthen the interfacial contact between active molecules and traditional materials, promoting their biocompatibility [82,83,84]. In addition, various structures and chemical compositions may trigger distinct osteogenic differentiation pathways such as bone morphogenetic proteins (BMPs), transforming growth factor-β (TGF-β) and Wingless-Type MMTV Integration Site Family (Wnt) signaling pathways to achieve robust osteogenic effects in the absence of exogenous osteoinductive growth factors (Fig. 1C) [85].
Clinically, surgical resection is frequently required to treat bone tumors, however, this procedure causes irreversible tissue damage, which has a substantial negative impact on patients’ quality of life [86]. Conventional chemotherapy and radiation are neither selective nor specific in destroying cells and can lead to major harmful side effects, so it is crucial to research more precise tumor-targeted therapeutic technique [87]. PTT uses heat produced by near-infrared (NIR) radiation to induce hyperthermia in tumor tissue, leading to protein denaturation, cell membrane rupture and subsequent death of cancer cells [88]. PDT kills cancer cells by generating singlet oxygen (SO) or reactive oxygen species (ROS) under light using photosensitizer molecules [89]. 2D nanomaterials with strong NIR absorption hold considerable promise for PTT/PDT of bone tumors, as demonstrated by numerous in vivo animal experiments that achieved positive therapeutic outcomes. Zeng et al. [19] effectively created (4-carboxybutyl) triphenyl phosphonium bromide (TPP)-conjugated indocyanine green (ICG)-loaded polyethylenimine-modified PEGylated nanographene oxide (PPG) (TPP-PPG@ICG), which can target mitochondria and trigger PTT and PDT during NIR irradiation, exerting a powerful killing impact on drug-resistant osteosarcoma cells both in vitro and in vivo (Fig. 1D). PTT based on 2D nanomaterials can also be used in treating bone metastases [90]. Ge et al. [91] used freeze-drying to polymerize GO nanoparticles, hydrated CePO4 nanorods and bioactive chitosan (CS) into CePO4/CS/GO porous scaffold that support bone regeneration and vascular development. Porous scaffolds were utilized for the postoperative treatment of bone metastasis induced by breast cancer. GO nanosheets effectively enhanced the photothermal conversion efficiency of photothermal therapy (PTT) in eradicating postoperative residual tumor cells, while CePO4 nanorods promoted angiogenesis and osteogenic activity to facilitate bone defect repair. The scaffold was able to raise the local temperature to 52 °C in mice within 30 s of near-infrared (NIR) irradiation. After two weeks of treatment, the tumor size in the treated mice was significantly smaller than that in the control group. In addition to act as a photothermal nanotherapeutic agent, GBN simultaneously transport anti-cancer medications to tumor sites for multi-pathway anti-cancer treatment (Fig. 1E) [92]. Zhang et al. constructed polyetheretherketone (PEEK)/graphene composite scaffolds using 3D printing and subsequently electrochemically deposited a hydroxyapatite-based, drug-loaded bioactive coating onto them. Graphene oxide (GO) loaded with the chemotherapeutic agent cisplatin served as an effective photothermal (PT) converter, and the scaffold was employed for multimodal cancer ablation therapy [92]. GO functioned as a PT converter within the scaffold, achieving a temperature of 45 °C within 100 s. At this elevated temperature, drug diffusion is enhanced, and near-infrared (NIR) irradiation further accelerates the release of the loaded drugs. In a mouse osteosarcoma model, implantation of the scaffold followed by NIR irradiation increased the local temperature above 45 °C within 100 s, maintaining approximately 50 °C throughout the remainder of the treatment period. After 10 days of treatment, the tumor weight was reduced by 98.5% compared to the control group. GO simultaneously functions as both a carrier for chemotherapeutic drugs and a photothermal agent, exerting a synergistic effect within this treatment platform through photothermal therapy (PTT)-mediated thermal damage to tumor cells and enhanced release of anticancer drugs at elevated temperatures, thereby improving its antitumor efficacy. Moreover, NIR laser induction can lower the dosage of chemotherapy drugs and boost the effectiveness of anti-cancer therapy [93].
To summarize, GBN exhibits remarkable potential in the field of BTE because to its exceptional honeycomb nanostructure, strong biocompatibility, coordinated biodegradability and mechanical stability as well as great cell-scaffold interaction capabilities. In the meantime, the light absorption characteristics and photothermal conversion efficiency brought by the unique electronic configuration together with targeted therapy bring hope to the management of bone tumors. The extensive chemical composition and diverse biological effects make GBN an attractive candidate for orthopedic medical applications.
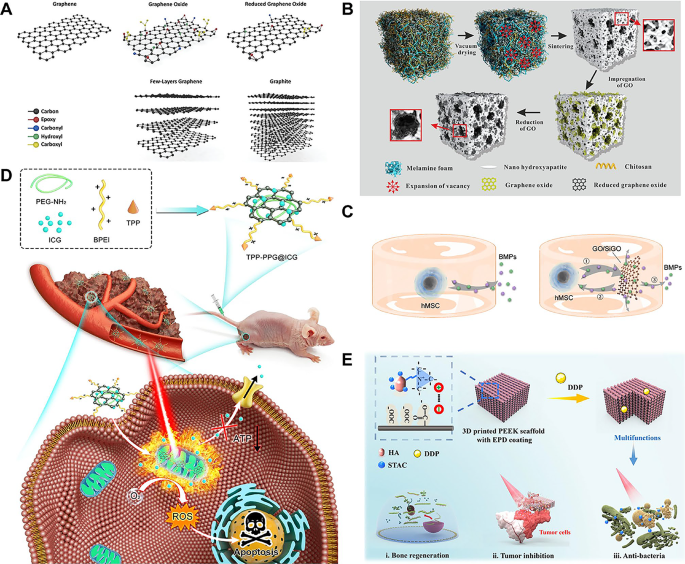
(A) Schematic illustration of various molecular structures of GBN. Reproduced with permission from Ref [63]. Copyright 2021, John Wiley and Sons. (B) Schematic diagram of the formation process of the porous HA/rGO scaffold. After the introduction of GO in porous HA ceramics, large GO sheets are attached to the surface of the through-hole structure, and small GO sheets are embedded in the hole wall. Reproduced with permission from Ref [80]. Copyright 2019, American Chemical Society. (C) The moderate binding affinity of BMPs to GO and SiGO nanosheets enables the dynamic deposition-release of endogenous BMPs produced by cells, creating a positive “feedback loop”. Reproduced with permission from Ref [85]. Copyright 2021, Elsevier. (D) Mitochondria-targeted TPP-PPG@ICG nanocomposites synergistically enhance antitumor efficacy after laser irradiation. Reprinted with permission from Ref [19]. Copyright 2021, Zeng et al. (E) PEEK/graphene nanocomposites loaded with antibiotics and anticancer drugs enable them to eradicate drug-resistant bacteria and ablate osteosarcoma cancer cells, and their therapeutic effects can be further enhanced by on-demand laser-induced heating. Reprinted with permission from Ref [92]. Copyright 2021, American Chemical Society. GBN, graphene-based nanomaterials; HA, hydroxyapatite; rGO, reduced graphene oxide; GO, graphene oxide; BMPs, bone morphogenetic proteins; TPP-PPG@ICG, (4-carboxybutyl) triphenyl phosphonium bromide-conjugated indocyanine green-loaded polyethylenimine-modified PEGylated nanographene oxide; PEEK, polyether ether ketone
BP
BP, also known as phosphoene, is a novel kind of 2D nano-layered semiconductor consisting of phosphorus atoms arranged in a honeycomb structure resembling graphene. In contrast to the planar structure of graphene, BP is an orthogonal rhombic structure with corrugates, formed by the covalent bonding of one phosphorus atom with three adjacent phosphorus atoms [94]. This crystal configuration allows lamellar BP to cause disruptions to cell membranes and display size-dependence [95]. Xing et al. synthesized black phosphorus (BP) nanosheets of varying sizes for evaluation. The average thicknesses of BP-1, BP-2, and BP-3 were 91.9 ± 32.0 nm, 27.0 ± 12.0 nm, and 17.0 ± 4.0 nm, respectively, while their lateral dimensions were 884.0 ± 102.2 nm, 425.5 ± 78.8 nm, and 208.5 ± 46.9 nm, respectively [95]. When co-cultured with cells in vitro, the smallest BP-3 showed the lowest level of cytotoxicity. Even at the highest concentration (200 µg/mL), BP-3 exhibited only moderate toxicity. The cytotoxicity of BP also varied among different cell types; for BP-1, the IC₅₀ value in the most tolerant HCoEpiC cells was over 30 times higher than that in the most sensitive 293 T cells. At exposure concentrations below 1 µg/mL, both BP-2 and BP-3 promoted cell proliferation to some extent, possibly due to the degradation of BP generating phosphates that stimulate cell growth. BP may also directly damage the cell membrane, resulting in the release of intracellular contents and subsequent cell death. The size of BP may influence its mode of interaction with the cell membrane; larger nanosheets may disrupt the integrity of the lipid bilayer, causing leakage of intracellular contents, while smaller nanosheets may deposit on the membrane surface, leading to minor disturbances. BP demonstrates significant anisotropy, with many of its properties dependent on the number of stacked layers supported by van der Waals forces [96].
Among the most stable allotropes of phosphorus, bulk BP was first synthesized over a century ago, while BP thin films were not discovered until 2014 [10]. Composed solely of phosphorus, a natural important constituent of human bone, BP-based biomaterials offer notable advantages in bone-related therapies [97]. BP exhibits excellent biocompatibility, as its constituent elements readily react with water and oxygen to degrade into non-toxic compounds such as phosphates and phosphites, which are easily excreted via the urinary system [98]. However, this chemical reactivity, coupled with a lack of mature synthetic strategies, has slowed the development of BP nanosheets in bone diseases despite its promising prospects. To achieve widespread application, the stability of BP can be enhanced through encapsulation, chemical modification, and doping with other elements [99]. Overall, BP-based biomaterials are primarily applied in BTE and bone tumor therapy [100].
In BTE aimed at repairing large bone defects, BP demonstrates robust osteogenic performance through a phosphorus-driven calcium capture process. Upon oxidation, BP degrades into phosphates that bind with surrounding free calcium ions to form calcium phosphate (CaP) crystal mineral deposits, thereby promoting in situ bone regeneration and repair of load-bearing tissues. Adequate calcium supplementation is beneficial for achieving an appropriate Ca/P ratio (Fig. 2A) [101]. Wang et al. [102] successfully synthesized double network and nano-engineered (NE) hydrogels, achieving adjustable mechanical features and a biomimetic extracellular matrix microenvironment. By incorporating multifunctional 2D BP nanosheets, NE hydrogels not only exhibit excellent mechanical properties but also induce CaP mineralization under mildly alkaline conditions, thus promoting bone tissue regeneration.
Furthermore, BP can leverage the inherent advantages of 2D nanomaterials by loading osteogenic or angiogenic drugs to achieve corresponding functionalities. BP nanosheets modified with bone morphogenetic protein-2 (BMP-2) facilitate the recruitment of osteoprogenitor cells and promote their differentiation into mature osteoblasts, thereby initiating the bone regeneration process (Fig. 2B) [103]. Liu et al. [104] constructed 2D heteronano-layers using BP and GO as the basis for functional spheroid design, aimed at providing a cell-guiding microenvironment for large bone defect repair. GO nanosheets enhance cell adhesion, migration, proliferation, and differentiation, while also improving the mechanical properties of biomaterials. In contrast, BP nanosheets possess an important characteristic for orthopedic applications: their degradation products are phosphates, which are not only harmless to the human body but also serve as essential ions for orthopedic and skeletal development, playing a crucial role in osteogenesis and bone integration. The combination of these two nanosheets can complement each other’s advantages. In this composite material, bone marrow mesenchymal stem cell spheroids are used to directly deliver stem cells to the defect site, reducing the time required for cell recruitment and accelerating the regeneration process. In both in vitro and in vivo experiments, these engineered spheroids were shown to promote stem cell proliferation and osteogenic differentiation, demonstrating their osteogenic support in a rat calvarial defect model. Additionally, by loading the osteogenic inducer dexamethasone, these scaffold-free spheroids achieved enhanced osteogenic induction without prior cultivation in osteogenic medium. Angiogenesis provides essential nutrients and oxygen, promoting the growth and repair of new bone tissue. The involvement of the nervous system contributes to angiogenesis and osteogenesis by releasing neuropeptides and other signaling molecules, thus playing a crucial role in bone regeneration. In Miao et al.’s study, BP nanosheets loaded with vascular endothelial growth factor (VEGF) enabled dynamic DNA hydrogels to be combined with 3D-printed scaffolds, constructing a bioactive gel scaffold structure. VEGF non-covalently bound with BP maintained sustained release, stimulating the migration of human umbilical vein endothelial cells (HUVECs), thereby providing adequate vascularization for bone defect repair (Fig. 2C) [105]. Xu et al.’s research demonstrated an innovative neurovascularized bone regeneration strategy through the design of a bilayer hydrogel. The upper layer of Mg-coordinated BP hydrogel mimicked the structure of the periosteum, significantly promoting angiogenesis by enhancing endothelial cell migration and neural-related protein expression in neural stem cells. The lower hydrogel layer effectively enhanced the activity and osteogenic differentiation of BMSCs. This study also validated the bone regeneration effect of the bilayer hydrogel scaffold in a rat calvarial defect model, indicating that this strategy offers a novel approach for designing neurovascular network biomaterials for bone regeneration (Fig. 2D) [106]. Although the degradation products of BP are non-toxic and promote osteogenic differentiation, BP nanosheets exhibit poor stability under aqueous and oxygen-rich conditions. Oxygen molecules on the surface of BP generate superoxide ions, which react with the lone-pair electrons on BP to form oxidized phosphorus. In the presence of water, the oxidized phosphorus rapidly degrades into phosphates. If degradation occurs during the initial implantation phase or even prior to implantation, it may negatively impact the final therapeutic outcome. Therefore, enhancing the stability of BP is essential. Combining BP with other materials can help address the issue of rapid degradation. Additionally, such combinations can impart multifunctional properties to the composite material, enhancing bioactivity while also providing additional functional characteristics. In orthopedic applications, BP also faces limitations in mechanical properties, as materials composed solely of BP cannot provide sufficient mechanical support for bone regeneration. Therefore, in the treatment of orthopedic diseases, BP is often combined with scaffold materials. The primary function of scaffold materials is to provide mechanical support, ensuring that the mechanical properties of the composite align with those of bone tissue to prevent premature collapse and stress shielding. Scaffold materials can also incorporate other components, integrating multiple complex properties to dynamically regulate bone regeneration in both time and space. Combining BP with scaffolds is an indispensable strategy for advancing the clinical translation of BP-based materials [98].
As a direct-bandgap, metal-free semiconductor, BP exhibits exceptionally high efficiency and performance in photoelectric conversion. Moreover, the bandgap of BP can be tuned according to the number of layers (ranging from 0.3 to 2.0 eV), facilitating broad-spectrum light absorption from visible to mid-infrared, thereby filling the bandgap void in this range left by graphene and TMDs [107]. Tong et al. [108] demonstrated that under NIR irradiation, BP exhibits an efficient photothermal response, significantly heating even when covered by biological tissues up to 7 mm thick. The photothermal properties of BP present distinct advantages in treating infectious bone defects. Jing et al. [109] proposed a novel strategy for infectious bone defects by utilizing a photosensitive conductive hydrogel for treatment. This hydrogel combines the antibacterial properties of magnesium-modified black phosphorus with photothermal/photodynamic therapeutic effects, effectively improving the infection-induced microenvironment and promoting the reconstruction of neural networks and bone regeneration. The study results indicated that this hydrogel exhibited excellent antibacterial performance under NIR irradiation, reducing the inflammatory response. This innovative strategy offers new directions for treating infectious bone defects, emphasizing the crucial role of nerves in bone repair, and provides potential new avenues for future therapies. Inspired by natural chloroplasts, Zhao et al. [110] developed a scaffold with photothermal stability and antibacterial properties. By inducing a multifunctional coating of polydopamine, BP was successfully stabilized in the scaffold, with the simultaneous growth of silver nanoparticles. This scaffold not only captures NIR light and converts it into thermal energy but also achieves pH-responsive release of silver ions and enhanced photothermal stability.
Nearly all types of bone defects undergo an inflammatory response as an initial stage, encompassing an early acute pro-inflammatory reaction followed by later inflammation resolution. This process is primarily utilized for cell recruitment, clearance of necrotic tissue, secretion of cytokines and growth factors, and promotion of cell differentiation. Macrophages, as the most common effector immune cells during the inflammatory phase, activate osteogenic signaling pathways and participate in all stages of bone healing [111]. Qiu et al. [112] revealed the importance of accelerating bone regeneration by modulating the inflammatory process and proposed a novel BP-based therapeutic strategy. The study demonstrated that BP could enhance the inflammatory response in the early stage and promote inflammation resolution in the subsequent stage, thus creating an immune microenvironment conducive to bone regeneration. Additionally, BP directly promotes osteogenic differentiation of BMSCs by stimulating macrophage-mediated interleukin-33 (IL-33) expression, further facilitating bone repair. This study provides new evidence for using BP to modulate immune responses to accelerate bone regeneration and offers potential strategies for future clinical treatments. Wu et al. [113] discovered that multifunctional hydrogels loaded with BP nanosheets exhibit excellent NIR/pH dual-responsive properties, which not only promote osteogenesis and angiogenesis but also demonstrate the ability to scavenge excessive ROS and induce macrophage polarization towards the M2 phenotype. Moreover, this hydrogel platform can drive the secretion of functional cytokines, accelerating bone regeneration.
Given their high biocompatibility, biodegradability, photothermal efficiency and drug-loading capacity, BP nanosheets have also been extensively studied for bone tumor therapy and anti-infection engineering (Fig. 2E) [114,115,116,117,118]. Zhao et al. [119] successfully developed a biomineralization-inspired CS/hydroxypropyltrimethyl ammonium chloride chitosan/HA/BP (CS/HC/HA/BP) hybrid scaffold for treating severe complications associated with bone tumors, such as tumor recurrence, infection and extensive bone loss. This scaffold possesses a bone-like hierarchical porous structure, enhancing bioactivity, osteoconductivity, and mechanical properties by regulating the HC/CS network matrix and in-situ crystallized HA nanoparticles distribution. The hybrid scaffold stabilized by HC exhibits good photothermal conversion properties, with the conversion temperature adjustable via BP concentration and NIR power density. In vitro and in vivo studies demonstrated the efficacy of a one-step NIR-mediated defense therapy, including antibacterial and antitumor effects, at approximately 49 °C. Subsequently, at around 42 °C, the scaffold promotes osteogenesis by upregulating the expression of osteogenesis-related genes. Wu et al. [120] prepared a promising antibacterial nanomedicine (ZnL2-BPs@HAP) by incorporating BP into the surface of 3D-printed HA scaffolds via ZnL2 coordination. ZnL2-BP not only imparts photothermal properties to the HA scaffold but also renders contacted bacteria more sensitive to high temperatures. Under conditions lower than conventional PTT temperatures, ZnL2-BPs@HAP scaffolds exhibited excellent antibacterial photothermal efficiency with minimal side effect. Furthermore, in another study, BP nanosheets (BPNS) effectively protected chondrocytes from ROS-induced degradation as a ROS scavenger and promoted osteogenic differentiation of BMSCs through its biomineralization capability, thereby achieving subchondral bone repair. In a rat OA model, BPNS showed effectiveness in preserving cartilage morphology and subchondral bone volume, making it a promising nanoplatform for early OA treatment [121].
In summary, BP-based nanomaterials represent a highly promising orthopedic implant material, addressing cancer treatment, antibacterial action and osteogenesis simultaneously. Their unique biological responses offer significant development potential in the field of bone therapy, and their combination with 3D printing substantially mitigates the shortcomings in fabrication techniques. BP-based nanomaterials are gradually becoming an ideal choice for orthopedic medical applications.
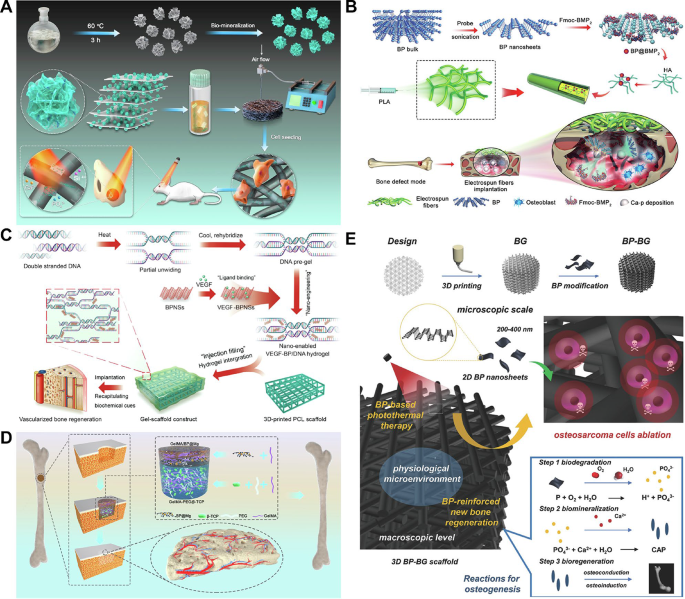
(A) Illustrating the fabrication of 3D BP@HA. BP nanosheets exert excellent photothermal properties and also provide a P source for bone regeneration through biomineralization. Reprinted with permission from Ref [101]. Copyright 2021, Li et al. (B) Schematic representation of the process of fabrication of BP@BMP-2 electrospun fiber scaffolds, as well as in vivo osteoblast recruitment and phosphate mineralization effects. Reprinted with permission from Ref [103]. Copyright 2020, John Wiley and Sons. (C) Schematic concept of bioactive gels for VEGF-modified BPNSs to enhance vascularized bone regeneration. Reprinted with permission from Ref [105]. Copyright 2022, Miao et al. (D) Diagrammatic illustration of a bilayer hydrogel scaffold that simultaneously induces neurovascular regeneration and osteogenesis. Reprinted with permission from Ref [106]. Copyright 2022, Xu et al. (E) Schematic diagram of a 3D printed BP-BG scaffold for photothermal therapy and subsequent bone regeneration for osteosarcoma. Reprinted with permission from Ref [114]. Copyright 2018, John Wiley and Sons. 3D, three dimensions; BP, black phosphorus; BMP-2, bone morphogenetic protein-2; VEGF, vascular endothelial growth factor; BPNP, BP nanosheets; BG, bioactive glass
Nanoclays
Nanoclays are natural or synthetic layered silicate particles composed of tetrahedral sheets (SiO4) connected with octahedral sheets [11]. Based on their chemical composition and nanoparticle morphology, nanoclays could be classified into several types, such as smectite (montmorillonite, saponite), kaolinite, pyrophillite and halloysite nanotube [15]. Laponite is a representative with formula expressed as Na+ 0.7 [Si8 Mg5.5 Li0.3O20(OH)4]−0.7, which has a permanent negative charge dispersed across the surface of the particle coupled with a positive charge along the edges [122]. The disc-shape morphology of nanoclays provides a high specific surface area for the attachment of biomolecules, and the dual charges distribution facilitates interactions at the edge sites of these biomolecules. One study has shown that bisphosphonates functionalized at the edge sites of laponite help maintain the local concentration of active proteins in vivo, thereby enabling bone induction at significantly lower doses of BMP-2 than usually required [123].
Nanoclays exhibit high biocompatibility, primarily because most of them consist of minerals naturally present in the body and degrade under physiological conditions into non-toxic products that can induce osteogenic differentiation. For instance, Mg2+ and Li+ participate in activating osteogenic regulatory pathways and promoting new bone formation, while Si and Si(OH)4 play critical roles in angiogenesis and the calcification of bone tissue during the regeneration process [124,125,126]. In addition to direct contact, nanoclay-based scaffolds are widely applied in bone and cartilage tissue engineering [127]. Firstly, the incorporation of nanoclays enhances the mechanical strength and degradation properties of composite scaffolds, yielding porous structures with mechanical characteristic similar to native bone matrices (Fig. 3A, B) [128, 129]. Hu et al. [130] significantly strengthened the mechanical and biological performance of methacrylated gelatin (GelMA) hydrogels by incorporating laponite nanoclays, creating a stable microenvironment that supports cell proliferation and differentiation. Moreover, the nanoclays permitted the controlled release qualities of hydrogel, allowing human umbilical cord-derived mesenchymal stem cell-derived small extracellular vesicles (hUC-MSCs-sEVs) to be gradually released as the hydrogel degraded, which in turn encouraged cartilage regeneration via activation of the PTEN/AKT signaling pathway. In vivo experiments confirmed the effectiveness of this GelMA/nanoclay/sEV hydrogel in repairing cartilage defects. The incorporation of nanoclay into the hydrogel significantly enhanced its resistance to degradation. After 14 days, the hydrogel was completely degraded, while the nanoclay-reinforced hydrogel exhibited only ~ 20% degradation and demonstrated enhanced mechanical properties. In a similar study, Yin et al. [131] introduced glycine-modified attapulgite (GATP), a natural nanometer-scale magnesium silicate mineral, into poly(1,8-octanediol-co-citrate) (POC) scaffolds, facilitating their compressive strength and biocompatibility while creating a conducive microenvironment for both cartilage and subchondral bone regeneration. The inclusion of GATP neutralized the acidity of citrate, reduced early inflammatory responses, and significantly promoted osteochondral differentiation. In a rabbit articular cartilage defect model, the POC/GATP composite scaffolds demonstrated excellent ECM secretion and osteochondral regeneration capabilities. Secondly, nanoclays act as bioactive additives that can promote cellular functions, encouraging cell adhesion, proliferation, and differentiation, sometimes achieving these outcomes without the need for additional growth factor or stem cell delivery (Fig. 3C) [132, 133]. The degradation behaviour of the POC/GATP composite scaffold in phosphate-buffered saline (PBS) solution is considered ideal, showing a mass loss of approximately 22.48% after an 8-week immersion period. Additionally, it exhibits a compressive modulus comparable to that of articular cartilage. In vivo experiments demonstrated that the POC/GATP scaffold exhibited repair effects comparable to those of natural cartilage. However, these biological processes are currently supported only by gene-level data, and investigations into the long-term stability and degradation behaviour in vivo remain incomplete. Future research should prioritize these performance aspects. Lee et al. [134] developed a nanoclay-organic hydrogel bone sealant (NoBS) by co-assembling inorganic silica-rich nanoclays with phytochemical-modified CS to mimic the extracellular microenvironment (ECM) of bone tissue. In this system, nanoclays not only enabled the osteoconductivity and physical characteristics of the hydrogel, but also promoted osteoinduction by regulating the Wnt/β-catenin signaling pathway. With the added benefits of antibacterial, antioxidant and self-healing features, NoBS has emerged as a promising alternative for bone defect repair, demonstrating considerable clinical potential (Fig. 3D). Thirdly, the presence of nanoclays can foster cell-material interactions through surface modification and functionalization, creating a favorable microenvironment for osteoblast behavior. High-throughput transcriptome sequencing has revealed that after cellular endocytosis, nanoclays activate stress response pathways, stimulating the differentiation of hMSCs towards osteogenic and chondrogenic lineages [135]. Zheng et al. [136] fabricated a laponite-functionalized 3D bioactive glass (BG) scaffold, where the incorporation of laponite significantly boosted the mechanical properties of the scaffold and improved osteogenic differentiation of adipose-derived stem cells (ADSCs). Under simulated hypoxic setting, the expression of hypoxia-inducible factor-1α (HIF-1α) and VEGF was upregulated, resulting in enhanced angiogenesis and bone regeneration both in vitro and in vivo (Fig. 3E). Despite these advantages, nanoclay still faces limitations regarding its long-term stability and degradation behaviour in vivo. The primary challenge lies in the mismatch between its degradation rate and the rate of bone tissue regeneration. This temporal and spatial mismatch not only weakens the mechanical support function of the scaffold but may also cause collapse at the bone defect site, ultimately compromising the efficacy of bone regeneration therapy. Due to complex and multifaceted degradation mechanisms, nanoclay struggles to synchronize its degradation with other materials, potentially resulting in premature scaffold collapse or localized stress concentration, thereby leading to failed bone repair. Additionally, degradation products of nanoclay may cause long-term biocompatibility concerns, as nano-sized silicate fragments produced during degradation can accumulate in organs such as the liver and spleen, thus affecting biosafety. The weakly alkaline microenvironment that may form locally during degradation could also affect osteoclast activity, interfering with mineralization and osteogenesis of the extracellular matrix. Limitations in manufacturing processes may further influence the degradation behaviour of nanoclay materials in vivo. Insufficient dispersion uniformity during manufacturing can lead to local aggregation of nanoclay particles, disrupting uniform degradation and ultimately resulting in unstable mechanical properties or scaffold collapse.
Through cation exchange, electrostatic adsorption, hydrogen bonding and cation bridging, nanoclays could readily bind to biomolecules such as proteins, providing a sustained release of various important growth factors and drugs to the cellular microenvironment [137]. Due to their flat, negatively charged sides, layer-by-layer films are formed via the parallel adsorption of flake laponite onto positively charged surfaces. Furthermore, the disc-like morphology of laponite impedes macromolecular diffusion, creating a tortuous path that significantly slows the release rate of loaded molecules between the layers. Based on this structure, Howard et al. used low-dose BMP-2 to significantly form well-structured Haversian systems in a rat calvarial defect model [138].
As a novel class of biomaterials, nanoclays possess unique layered morphology and charge distribution, allowing them to interact with scaffold materials, cells and ECM components. Nanoclays are easily synthesized and cost-effective, with controllable sizes and defined structures when used as precursor materials for 3D bioprinting, responding to bone induction signaling pathways [139]. Nanoclay interacts with cells and biomolecules within the osteogenic microenvironment to exert osteogenic effects. Magnesium ions (Mg2+) and silicon ions (Si4+) released from nanoclay activate osteogenic-related signalling pathways, including BMP/Smad and Wnt/β-catenin, which promote the activation of osteogenic-specific transcription factors such as Runx2, Osterix, and osteopontin, thereby inducing the differentiation of bone marrow mesenchymal stem cells (BMSCs) into osteoblasts [140]. Silicon ions also promote angiogenesis coupled with osteogenesis by stabilizing hypoxia-inducible factor 1α (HIF-1α), thereby activating the VEGF axis and providing nutritional support for bone regeneration [141]. The negatively charged surfaces of nanoclay can adsorb calcium and phosphate ions through electrostatic interactions, thereby forming mineralization precursors and accelerating bone matrix mineral deposition. Additionally, montmorillonite inhibits osteoclast differentiation and bone resorption by downregulating the expression of key genes, including NFATc1 and TRAP [142].
Nanoclays can also bind biomolecules via electrostatic interactions, enabling controlled release of growth factors. For example, the negatively charged layers of nanoclays can bind to the positively charged amino acid domains of bone morphogenetic protein-2 (BMP-2), while their hydrated surface layers help maintain BMP-2’s native conformation, thereby preserving its bioactivity and enabling sustained release. In the complex bone microenvironment, the targeting specificity of nanoclay can be enhanced via surface modification. Conjugation of targeting ligands, such as bisphosphonates or RGD peptides, enables nanoclay to specifically target bone minerals or osteoblasts. Additionally, biomimetic mineralization strategies can be employed by in situ deposition of hydroxyapatite on the nanoclay surface to mimic natural bone tissue, thereby enhancing bone conductivity. Controlled drug release from nanoclay carriers can utilize multiple response mechanisms, including protonation of nanoclay in acidic environments to trigger drug release, or enzyme-responsive strategies targeting specific enzymes in the bone microenvironment, such as matrix metalloproteinases (MMPs) [143, 144].
In designing bone-based composite materials using nanoclay, optimizing the concentration and spatial distribution of nanoclay is critical to achieving a balance between mechanical properties and biological activity. The addition of nanoclay imparts bioactivity to the composite material. At low concentrations, nanoclay can be uniformly dispersed within the matrix, enhancing the compressive modulus of the scaffold via non-covalent interactions, including hydrogen bonding and electrostatic forces, while maintaining high porosity to facilitate nutrient transport and cell migration. However, excessive nanoclay can lead to stress concentration and heterogeneous degradation rates due to agglomeration, thereby impairing osteogenic performance. For example, Yin et al. [131] reported that the addition of GATP effectively increased the compressive modulus of POC scaffolds, with a 10% addition approaching that of articular cartilage. However, when the GATP concentration increased to 15%, the compressive modulus of the scaffold decreased, as excessive GATP filled small pores, reducing internal connectivity and resulting in uneven stress distribution within the scaffold under compression. Strategies such as surface modification or dynamic cross-linking network design can mitigate limitations associated with nanoclay concentration. Regarding the spatial distribution of nanoclay, it must align with the gradient structure of bone-cartilage tissue, characterized by lower mineralization in the cartilage layer and higher mineralization in the bone layer. Programmable control of nanoclay density can be achieved through techniques such as gravity permeation, 3D printing, hot-press curing, and multi-material co-assembly to meet the mechanical load requirements of various anatomical regions. The key to regulating nanoclay concentration and distribution lies in real-time synchronous monitoring of parameter changes and optimization of manufacturing processes, which is also critical for scalable nanoclay production [145].
Future efforts should prioritize the development of clinically relevant animal models, establish manufacturing standardization (including GMP-compliant production processes), and conduct preclinical studies that comply with regulatory requirements to supplement in vivo data on the long-term biosafety, systemic clearance, and inflammatory responses of nanoclay, as well as its interactions with complex human tissues, particularly under pathological or immunocompromised conditions. Additionally, efforts should focus on integrating nanoclay into bio-manufacturing platforms, such as 3D bioprinting and injectable hydrogels, to enhance spatiotemporal control over drug delivery and tissue formation.
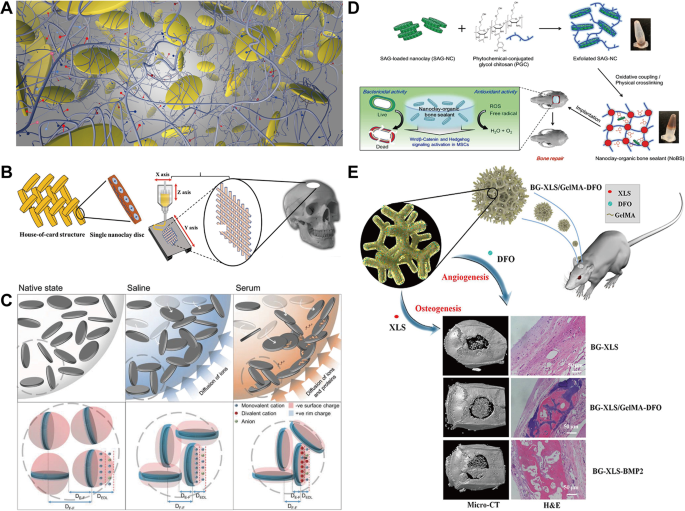
(A) Schematic drawing of the formation of a composite hydrogel by covalent crosslinking of nanosilicate and GelMA under ultraviolet irradiation. Reprinted with permission from Ref [128]. Copyright 2015, American Chemical Society. (B) Laponite with negatively charged surfaces and positively charged edges can form a “house of cards” structure through edge-to-edge interactions. Laponite using 3D bioprinting improves the mechanical strength of bone implants. Reprinted with permission from Ref [129]. Copyright 2019, John Wiley and Sons. (C) Diagrammatic presentation of the mechanism by which Laponite diffusion gels are formed in different liquid environments. Reprinted with permission from Ref [132]. Copyright 2018, John Wiley and Sons. (D) Schematic diagram of the co-assembly of inorganic nanoclay and organic phytochemically enhanced chitosan to achieve self-healing, antibacterial and antioxidant activities. Reprinted with permission from Ref [134]. Copyright 2020, John Wiley and Sons. (E) Laponite-functionalized BG scaffold with hypoxic properties that promotes angiogenesis and osteogenesis. Reprinted with permission from Ref [136]. Copyright 2021, Zheng et al. GelMA, methacrylated gelatin
LDHs
LDHs, also known as anionic clays, are compounds composed of positively charged layers and interlayer anions stacked together via electrostatic interactions. Their general formula is [M1−x2+Mx3+(OH)2]x+(An−)x/n·mH2O, where M2+ and M3+ represent divalent and trivalent metal cations respectively and An− refers to exchangeable interlayer anions [23]. The metal ions within the layers of LDHs display remarkable tunability, with distinct metal ions mediating specific pathophysiological processes. For instance, Mg-LDHs facilitate the proliferation of osteoblasts and exhibit superior anticorrosion properties [146]. Zn-LDHs and Cu-LDHs mitigate oxidative stress through anti-inflammatory and antioxidant qualities, thereby enabling antimicrobial and antitumor immunity (Fig. 4A) [147,148,149]. Also, europium-based LDHs enhance vascularization and promote the repair of both soft and hard tissues (Fig. 4B) [150].
LDHs have garnered extensive attention in the field of pharmaceuticals delivery. Their intrinsic positive charge encourages non-covalent interactions with anionic drugs, biomaterials and antibodies, substantially boosting transmembrane internalization and intracellular drug delivery efficiency. Furthermore, the presence of hydroxides imparts acid sensitivity to LDHs, rendering them ideal candidates for pH-responsive drug carriers in cancer therapy. Drug-loaded LDHs exhibit controlled release characteristics, whereby intercalated drugs are gradually liberated, reducing the frequency of administration. Lv et al. [151] developed a smart, injectable, thermosensitive hydrogel (CSP-LB) by incorporating BMP-2-functionalized MgFe-LDH nanosheets into a chitosan/silk fibroin (CS) hydrogel loaded with platelet-derived growth factor-BB (PDGF-BB). In this hydrogel system, the relatively weak interaction between PDGF-BB and the CS hydrogel, in contrast to the strong electrostatic interaction between BMP-2 and the LDH nanosheets, enabled rapid PDGF-BB release coupled with sustained BMP-2 release, thus achieving efficient and prolonged bone regeneration.
In the realm of BTE, the integration of LDHs not only increases the bioactivity of inorganic matrices, promoting osteoblast adhesion and proliferation, but also serves as a coating on metallic substrates. Such coatings effectively retard in vivo degradation, improving mechanical stability. For example, Wang et al. [152] demonstrated that MgAl-LDHs effectively fostered the bioactivity and application potential of polymethyl methacrylate (PMMA) bone cement. LDH incorporation notably reduced the polymerization temperature of PMMA, alleviating stress shielding effects while promoting robust bone cement interfacial integration and markedly enhancing bone formation in vivo. Similarly, studies revealed that MgAl-LDHs on pure magnesium surfaces successfully induced the differentiation of osteoblasts (MC3T3-E1) and the angiogenic capability of HUVECs. Additionally, LDH coatings induced macrophage polarization toward the anti-inflammatory M2 phenotype, optimizing the local immune microenvironment and suppressing the activation of the NF-κB signaling pathway (Fig. 4C) [153]. Zhang et al. [154] fabricated Mn-LDH nanosheet-modified magnesium-based implants, discovering that the black LDH film, through its superior photothermal effect and nanozyme-like Fenton activity, efficiently eradicated osteosarcoma cells and tissues under NIR light while simultaneously exerting photothermal antibacterial capacities. Moreover, the LDH film greatly raised the corrosion resistance of magnesium-based implants, precisely controlled the release of Mg2+ and Mn2+, and synergistically stimulated cell adhesion, proliferation and osteogenic differentiation, thereby accelerating bone regeneration. A novel study reveals that LDH coatings can be tailored across multiple scales to match the structural and functional requirements of 3D-printed magnesium scaffolds, ensuring both biocompatibility and controlled degradation. At the macroscopic scale, the scaffold exhibits an anatomically precise geometry, enabling seamless integration with bone defects. At the mesoscale, an optimized porous architecture provides mechanical support while facilitating cellular proliferation and vascularization. At the microscale, a dual-layer coating comprising a high-temperature oxidation (HTO) film and an LDH layer establishes a protective barrier, mitigating rapid magnesium degradation while modulating the microenvironment to enhance osteogenic activity. At the nanoscale, LDH nanosheets improve surface roughness and hydrophilicity, thereby promoting cell adhesion and differentiation. The synergistic interplay across these hierarchical levels ensures structural integrity while accelerating bone tissue regeneration, offering a promising avenue for customized bone repair solutions [155].
The antimicrobial features of LDHs have also been extensively investigated. By intercalating ciprofloxacin and gentamicin into the interlayer space of MgAl-LDHs, Cámara-Torres et al. [16] successfully achieved sustained local antibiotic release within 3D-printed scaffolds to prevent implant-associated bone infections. Importantly, these scaffolds supported bone tissue formation without impairing the osteogenic differentiation or marker expression of hMSCs, demonstrating excellent biocompatibility (Fig. 4D). In addition, Bian et al. [156] developed a BG-based composite (BGS/MCFS) functionalized with sulfide nanosheets derived from MgCuFe-LDHs, addressing prosthesis-associated infections and loosening. The antibacterial efficacy of LDHs primarily stems from their inhibition of bacterial energy production and metabolism. Combined with exceptional NIR photothermal conversion abilities, BGS/MCFS significantly elevated temperatures under irradiation, enabling complete eradication of bacteria surrounding implants.
In conclusion, LDH-based nanomaterials offer a highly tunable and multifunctional platform for orthopedic applications, capable of integrating osteoinductive, angiogenic, anti-inflammatory and antimicrobial properties into a single construct. Through rational selection and combination of metal ions, interlayer engineering and responsive drug loading, LDHs can be tailored to address diverse clinical scenarios from enhancing osseointegration to combating implant-associated infections and supporting tumor resection margins.
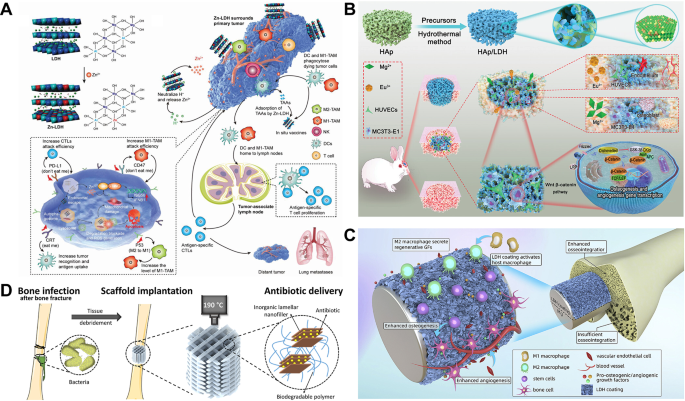
(A) Zn2+-doped LDH for tumor microenvironment regulation and tumor-specific immune induction. Reprinted with permission from Ref [148]. Copyright 2022, John Wiley and Sons. (B) Schematic illustration of in situ growth of MAE-LDH nanosheets on porous HA scaffolds. The composite material owns excellent angiogenic and osteogenic properties. Reprinted with permission from Ref [150]. Copyright 2022, Wang et al. (C) MgAl-LDH coating promotes immune response of magnesium implants and exhibits better bone integration performance. Reprinted with permission from Ref [153]. Copyright 2020, Cheng et al. (D) Antibiotic-loaded 3D printed MgAl-LDH scaffolds for bone regeneration and infection prevention. Reprinted with permission from Ref [16]. Copyright 2020, Cámara-Torres et al. LDH, layered double hydroxides; MAE-LDH, MgAlEu-LDH
TMDs
2D TMDs, which share a similar structure to graphene, consist of MX₂-type crystals, where M represents transition metals from groups IV to X (Mo, W, V and Ti) and X denotes chalcogenides (S, Se and Te). In each monolayer, TMDs feature a “sandwich-like” construction with a layer of metal atoms sandwiched between two layers of chalcogen atoms, bonded covalently within the layer. Vertically, adjacent layers are held together by weaker van der Waals forces, allowing for facile exfoliation into 2D nanosheets without disrupting the internal bonding [157]. Platinum diselenide (PtSe2), in its bulk form, exhibits an indirect bandgap, but transitions to a direct bandgap semiconductor when flaked into a monolayer, making it highly suitable for applications in photosensitive drug delivery and PTT (Fig. 5A) [158, 159].
The crystal phase is a critical determinant of the properties and functionalities of TMDs, and phase engineering allows for precise modulation of crystal phase transitions, thus endowing TMDs with multifaceted chemical characteristics [160]. Molybdenum disulfide (MoS₂), for example, exists in four distinct crystalline structures, namely 2H, 1 T, 1T’, and 3R, with the semiconductor 2 H phase and metallic 1 T phase being the most prevalent [161]. The 2 H phase represents the thermodynamically stable form of MoS₂ and, when stimulated by light or electrons, increases osteoblast proliferation and differentiation while maintaining excellent biocompatibility. Conversely, the metallic 1 T phase, obtained from 2 H via chemical treatments such as intercalation of alkali metal lithium, exhibits superior conductivity, heightened catalytic activity and enhanced antibacterial properties, rendering it advantageous for antitumor therapies (Fig. 5B) [159, 161,162,163].
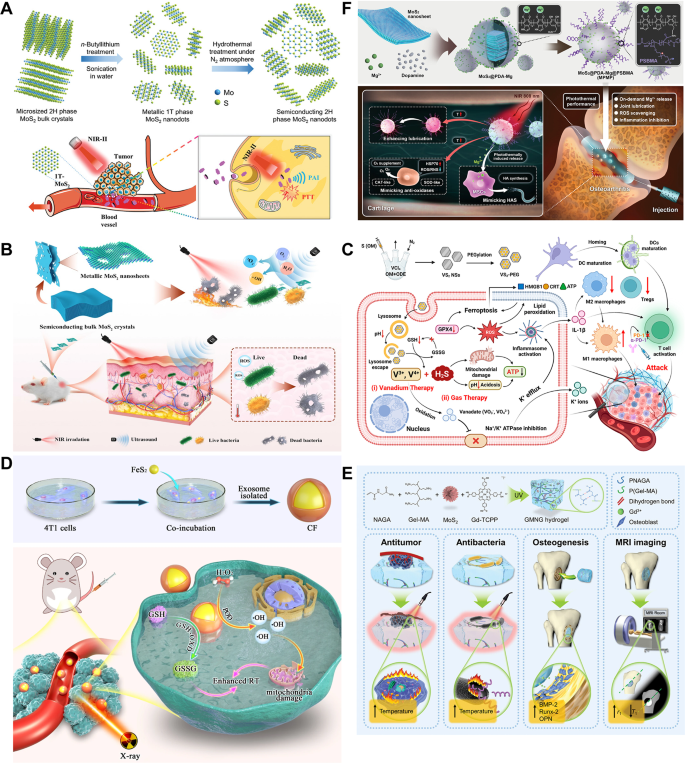
By adjusting synthesis methods and process parameters, different crystalline phases of transition metal dichalcogenides (TMDs) can be obtained. Mechanical exfoliation, the earliest physical method, suffers from weak interlayer interactions, resulting in loosely stacked layers. Ion intercalation and exfoliation use ions that react with water to generate gas, thereby separating the layers; lithium ion intercalation, in particular, allows precise control over the key structure of TMDs. Additionally, liquid-phase exfoliation and chemical vapor deposition are commonly employed production techniques. Optimizing these synthesis processes enables phase control of TMDs [164]. Park et al. developed a melt-metal-assisted intercalation (MMI) strategy leveraging the capillary action of molten potassium and the difference in electron affinity between MoS2 and potassium’s ionization potential. The highly reactive molten potassium readily intercalates into MoS2 interlayers, lowering the energy barrier for the 1 T phase transition and stabilizing the 1 T crystal structure with phase purity exceeding 92%. The suppression of electron emission by K–S ionic bonds imparts excellent thermal and photostability [165]. High-temperature treatment of alkali metal salts in reducing atmospheres can also drive phase transitions, where the salts act to lower energy barriers and provide electron doping. Furthermore, colloidal thermal injection technology allows direct growth of single-layer 1T’-TMD crystals by regulating ligand supersaturation with precursors [166]. Chen et al. prepared 1T/1T’-MoS2 via chemical lithium intercalation exfoliation by immersing 2 H-MoS2 in n-butyl lithium solution to induce phase transformation, followed by sonication in water to obtain water-dispersible MoS2. They also demonstrated that simple hot water treatment under nitrogen atmosphere can revert 1T/1T’-MoS₂ back to the 2 H phase.
Different crystalline phases of MoS2 exhibit distinct properties, making phase purity critical for the reproducibility and efficacy of TMDs in orthopaedic applications. Mutalik et al. investigated the optical differences between the 2 H and 1 T phases of MoS2: 1T-MoS2, due to its metallic nature, shows higher absorbance at 808 nm, while 2 H-MoS2 exhibits very low absorption in the near-infrared window owing to its wide bandgap (~ 1.83 eV). Regarding photothermal performance, the 1 T phase converts near-infrared light to heat more rapidly and efficiently, reaching temperatures above 70 °C within 7 min, significantly outperforming the 2 H phase, which only reaches 50 °C. In antimicrobial experiments, 1T-MoS2 effectively reduced E. coli populations by causing extensive damage to bacterial cell walls and disrupting growth, attributed to its metallic conductivity enhancing bacterial toxicity and photothermal effect [167]. Chen et al. further confirmed the superior photothermal effect of 1T/1T’-MoS2 compared to 2 H-MoS2 under 1064 nm laser irradiation: the 1T/1T’-MoS2 solution reached 58.9 °C within 3 min, whereas the 2 H-MoS2 solution reached only 35.8 °C. As a sonosensitizer, 1T/1T’-MoS2 generated approximately 45% more reactive oxygen species (ROS) under ultrasonic treatment than 2 H-MoS2. In antibacterial assays, 1T/1T’-MoS2 achieved bactericidal rates of 18% and 20% against Pseudomonas aeruginosa under ultrasonic and light treatments respectively, with combined treatment reaching nearly 100% efficacy. These results were corroborated in vivo. However, Chen et al. noted that the purity of 1T/1T’-MoS2 was still limited, with around 30% of nanosheets remaining as the 2 H phase [163].
Therefore, phase purity is essential for reproducible and effective TMD applications in orthopaedic disease treatment. It directly affects batch consistency and significantly influences physical and chemical properties such as surface energy and degradation rate, which in turn impact photothermal performance. The metallic 1 T phase’s zero-bandgap structure grants it strong light absorption and lower excitation energy thresholds, enhancing ROS generation upon activation, thereby making it superior for photothermal therapy. In contrast, the semiconducting 2 H phase may be better suited for promoting osteoblast differentiation via controlled ion release.
In the context of orthopedic disease management, TMDs demonstrate vast potential across multiple applications. MoS₂ nanosheets, characterized by their strong NIR absorption and high photothermal conversion efficiency, serve as ideal agents for PTT while also regulating cellular activities [168]. Additionally, the unique electronic structure of transition metals confers catalytic properties on TMDs, enabling them with exceptional peroxidase (POD)-mimetic activity. Vanadium disulfide (VS₂), iron disulfide (FeS₂), and molybdenum diselenide (MoSe₂) decompose hydrogen peroxide (H₂O₂) to generate ROS, such as hydroxyl radicals (•OH), which induce apoptosis and DNA damage in tumor cells (Fig. 5C, D) [169,170,171]. Huang et al. [172] manufactured a multifunctional hydrogel (GMNG), incorporating MoS₂ and gadolinium (Gd)-complex dopants into N-acryloyl glycinamide/Gel-MA (NAGA/Gel-MA), for osteosarcoma treatment. Through its high-efficiency photothermal conversion, MoS₂ eradicated tumor cells both in vitro and in vivo while preventing postoperative bacterial infections. This hydrogel system, capable of releasing Gd³⁺, simultaneously promoted osteogenesis and enabled magnetic resonance imaging (MRI)-based monitoring of its degradation, offering advanced strategies for bone defect repair (Fig. 5E).
In BTE, 2D TMDs are frequently employed as surface coatings to create a microenvironment conducive to osteoblast growth [173]. Furthermore, defect-rich MoS₂ nano-assemblies provide abundant active sites, serving as crosslinking centers that modulate the kinetics and mechanical characteristics of polymer hydrogels, resulting in stronger networks [174]. Ma et al. [175] established an innovated electrospun poly(ε-caprolactone)/MoS₂ (PCL/MoS₂) composite membrane for guided bone regeneration. MoS₂ nanosheets, acting as both osteogenic promoters and NIR photothermal agents, supplied the composite membrane with outstanding photothermal performance, stronger mechanical properties and maintained biodegradability. Under NIR irradiation, the PCL/MoS₂ composite membrane significantly boosted the proliferation of BMSCs and aided in the healing of rat tibial lesions compared to pure PCL membranes. Yuan et al. [176] proposed a functionalized MoS₂/PDA-arginine-glycine-aspartic acid (RGD) coating for Ti implant. Upon NIR rays, the MoS₂ nanosheets induced localized hyperthermia via the photothermal effect, promoting rapid glutathione oxidation and heightening bacterial susceptibility to oxidative stress. Concurrently, through a ROS-independent oxidative stress mechanism, the MoS₂ compromised bacterial membrane permeability, leading to protein leakage and adenosine triphosphate (ATP) depletion, effectively inhibiting bacterial proliferation. In addition, the MoS₂ coating raised cellular compatibility and osteogenic activity by upregulating the expression of osteogenesis-related genes, such as alkaline phosphate (ALP), runt-related transcription factor 2 (Runx2), collagen Type I (Col I) and osteocalcin (OCN). In vivo studies demonstrated that MoS₂/PDA-RGD-modified Ti implants, under NIR irradiation, not only efficiently eradicated Staphylococcus aureus (S. aureus) infections but also accelerated new bone formation, showcasing remarkable antibacterial and osseointegration-promoting efficiency.
Yu et al. [177] developed a novel biomimetic photothermal nanozyme (MPMP) based on MoS₂, doped with Mg²⁺-modified PDA and coated with amphiphilic polysulfobetaine, for the amelioration of OA. This nanozyme mimicked antioxidant enzymes/hyaluronan synthase, promoting cartilage regeneration, enhancing lubrication and scavenging ROS and reactive nitrogen species (RNS) under NIR irradiation, thereby mitigating inflammation. Research shows the MPMP nanozyme significantly suppressed the NF-κB/IL-17 signaling pathway while activating the MAPK pathway, facilitating cartilage repair and hyaluronic acid production, ultimately ameliorating OA symptoms in a mouse model (Fig. 5F). Moreover, another study highlighted the potential of MoS₂-incorporated polyether ether ketone (PEEK) composites in enhancing artificial joint materials. The solid lubricating effectiveness of MoS₂ significantly reduced friction and wear, while its layered structure enabled the formation of a stable transfer film under shear, reducing wear rates. MoS₂ also improved the compressive strength and hardness of the composite, maintaining excellent cellular compatibility and promoting BMSC proliferation, underscoring its biological advantages in joint prosthetics [178].
Despite these promising properties, TMD-based systems continue to face significant challenges in clinical translation. A major concern lies in the long-term biological safety of transition metals, particularly under conditions of chronic exposure and in metabolically sensitive environments like bone marrow. The degradation of TMDs results in the release of metal ions (e.g., M6+, W6+) and the metabolism of sulfur-containing products (e.g., sulfate ions, thiol compounds), leading to complex toxicological mechanisms. Yim et al. investigated the degradation pathways of TMDs in mice, reporting that at 3 h post-injection, tungsten dichalcogenide (WS2) showed distinct signals in the liver, spleen, lungs, and kidneys. Three days after intravenous injection, no fluorescent signals from the nanoparticles were detected in any of these organs, indicating rapid clearance and reduced long-term toxicity risk. However, following intraperitoneal administration, the nanoplates distributed more rapidly to major organs within 1 h, with trace signals still detectable in the liver and lungs at 10 days, suggesting prolonged retention compared to intravenous injection [179]. Xue et al. examined the tissue distribution of MoS2, finding that pure MoS2 nanosheets accumulated in the lungs within 1 h post-administration, whereas signals in the heart, kidneys, and brain were minimal. At 1 and 2 days post-administration, signal intensity in the spleen and liver increased significantly, possibly due to the size of MoS2 particles, as larger particles may have difficulty passing through the glomerular filtration barrier or crossing vascular barriers in the heart and brain. Thirty days following intravenous injection, a small quantity of MoS2 nanosheets remained detectable in the spleen, mainly localized within the red pulp region [180]. Different TMDs exhibit varying in vivo metabolic rates. One day post-administration, MoS2, WS2, and TiS2 showed similar accumulation in the liver and spleen, likely attributed to phagocytosis by Kupffer cells and splenic macrophages. Over time, MoS2 exhibited a significantly faster metabolic rate compared to WS2 and TiS2, with its in vivo levels markedly decreasing within one month post-administration. Elevated molybdenum concentrations detected in urine and feces suggest renal and intestinal excretion of MoS2, whereas WS2 and TiS2 display prolonged retention in the body. MoS2 degradation primarily yields water-soluble high-valent Mo(VI) oxides, while WS2 and TiS2 undergo less complete degradation. WS2 is not fully oxidized to W(IV)/W(VI) compounds, and oxidized TiS2 forms insoluble white TiO2 precipitates [181].
Regarding TMD biocompatibility, exfoliated 2D-MoS2 is generally considered to exhibit low cytotoxicity. However, these conclusions predominantly rely on basic cell viability assays and lack comprehensive evaluation of cell morphology, metabolism, or transcriptional responses. Some studies report conflicting results regarding MoS2 cytotoxicity, potentially due to variations in experimental design and material preparation. Variables including concentration, cell culture conditions, cell types, exposure parameters, nanosheet structure, and lateral size may influence these outcomes. Toxic effects broadly manifest as alterations in cell viability driven by oxidative stress or cellular internalization. In vivo, MoS2 accumulation in organs can induce inflammation and immune responses, contributing to toxicity. Although 3D in vitro cell culture models better mimic tissue environments for toxicity assessment, they cannot fully replicate systemic organismal responses [182]. The limited research on TMD toxicity raises concerns regarding potential long-term risks in orthopaedic applications. Ion release during TMD degradation may trigger a “metal ion release storm.” Prolonged accumulation of TMD degradation products may cause organ fibrosis, resulting in progressive impairment of hepatic and renal functions.
Moving forward, translational research should emphasize the development of biodegradable or ion-doped TMD derivatives that retain therapeutic efficacy while reducing systemic burden. Moreover, standardized models of bone infections, osteosarcoma, and osteoarthritis are needed to better simulate clinical conditions and evaluate long-term effects. Regulatory-oriented studies focusing on toxicology, clearance pathways, and immune interactions will be pivotal for enabling clinical adoption.
In summary, TMDs, with their distinctive structure and multifunctional features, hold immense promise in the treatment of orthopedic diseases. Through phase engineering, POD activity and photothermal effects, TMDs achieve microbial eradication, tumor ablation and bone tissue regeneration. As research progresses, TMDs are expected to become key materials in the development of innovative therapeutic approaches for orthopedic disorders.
(A) The schematic diagram shows the preparation of metal 1T-phase MoS2 to semiconductor 2 H-phase MoS2 nanodots from micron-scale 2 H-phase crystals. Among them, 1T-MoS2 nanodots can be used as a highly effective reagent for tumor PTT. Reprinted with permission from Ref [159]. Copyright 2020, John Wiley and Sons. (B) Schematic representation of metal 2D MoS2 nanosheets obtained by liquid peeling for photothermally enhanced acoustic antimicrobial. Reprinted with permission from Ref [163]. Copyright 2022, Chen et al. (C) Diagrammatic illustration of VS2-PEG preparation and its enhanced cancer immunotherapy. Reprinted with permission from Ref [169]. Copyright 2023, American Chemical Society. (D) FeS2 nanozymes combined with cancer cell-derived exosome therapy leads to radiation sensitization through an oxidation reaction, thereby reducing the radiation dose of radiotherapy. Reprinted with permission from Ref [171]. Copyright 2021, Huang et al. (E) The hydrogel doped with MoS2 nanoroses has the ability to kill tumors, prevent bacterial infections and promote the formation of new bones. Reprinted with permission from Ref [172]. Copyright 2023, John Wiley and Sons. (F) MoS2-based nanozyme-based composites can be used for photothermal therapy of osteoarthritis. Reprinted with permission from Ref [177]. Copyright 2023, John Wiley and Sons. 2D, two-dimensional; MoS2, molybdenum disulfide; PTT, photothermal therapy; VS2, vanadium disulfide; PEG, polyethylene glycol; FeS2, iron disulfide
MXenes
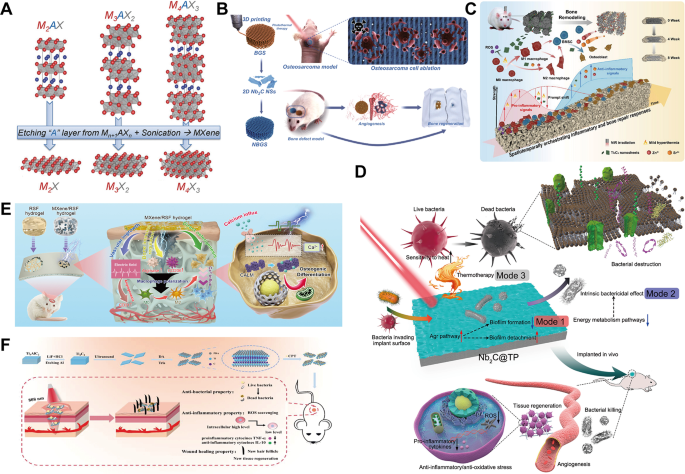
MXenes, a novel class of 2D nanomaterials with a layered topological structure, were first discovered in 2011 [183]. These materials, encompassing transition metal carbides, nitrides or carbonitrides, are represented by the general formula Mn+1AXn, where M denotes an early transition metal, A represents elements from groups 13 or 14 of the periodic table and X stands for carbon or nitrogen (Fig. 6A) [184]. Due to their excellent electrical conductivity, high specific surface area, tunable surface functional groups and chemical stability, MXenes have garnered significant attention in biomedical applications.
Among the degradation products of MXenes, certain transition metal ions are believed to possess osteogenic potential. For instance, Ti⁴⁺ ions can promote the proliferation and differentiation of osteoblasts, while niobium (Nb) ions enhance the adhesion and growth of these cells (Fig. 6B) [185, 186]. Studies have demonstrated that optimal levels of Ti⁴⁺ can activate inflammatory and osteogenic signaling pathways, such as the PI3K-Akt and ERK pathways (Fig. 6C) [186, 187]. Additionally, both Ti⁴⁺ and Nb, at low concentrations, exhibit antimicrobial activity, thus reducing the risk of postoperative infections (Fig. 6D) [188].
High electrical conductivity and biocompatibility of MXenes make them particularly suitable for supporting osteoblast proliferation and differentiation [189]. This property is especially advantageous in electrically stimulated bone repair, where MXenes significantly amplify osteogenic effects and increase bone regeneration [190]. For example, He et al. [191] synthesized a nanocomposite material combining MXene with a narrow-bandgap sonosensitizer (VS4) to construct a Schottky junction-based system (VSM). In this configuration, MXene played a pivotal role in augmenting the separation efficiency of electron-hole pairs and, through its superior conductivity and 2D electronic acceptor properties, markedly improved the sonodynamic and chemodynamic effects of VS4. This synergy enabled VSM to exhibit outstanding antibacterial activity under ultrasonic stimulation while enhancing osteogenesis through the stable release of vanadium ions. In another study, Hu et al. [192] developed a dual-crosslinked conductive hydrogel network by integrating MXene with regenerated silk fibroin (RSF). This hybrid system effectively transmitted electrical signals and activated the Ca²⁺/CALM signaling pathway in osteoblasts, thus strengthening their proliferation and differentiation. Moreover, the hydrogel improved the bone immune microenvironment and facilitated angiogenesis, underscoring its unique value as a conductive material for bone repair applications (Fig. 6E).
Ti₃C₂Tₓ, a representative MXene material, typically carries surface functional groups (Tₓ) such as -OH, -F, or -O, which arise from its exposure to acidic or alkaline solutions during synthesis [184]. These surface-active sites raise the water solubility and dispersibility of Ti₃C₂Tₓ while inhibiting bacterial growth through oxidative stress, making it a promising antimicrobial coating for orthopedic implants. Yu et al. [193] demonstrated that the in situ oxidation of Ti₃C₂, driven by CaO₂-mediated H₂O₂ release, generated TiO₂ sonosensitizers and catalytically active Ti³⁺. This process significantly boosted ROS production, thereby elevating antibacterial efficiency. Simultaneously, the release of Ca²⁺ ions facilitated mineralization at the bone defect site, supporting bone regeneration. Both in vitro and in vivo experiments revealed that composite nanosheets exhibited exceptional antibacterial and bone repair capabilities in prosthetic joint infection and osteomyelitis models. Furthermore, MXenes can act as carriers for antibiotics or antimicrobial peptides, enabling the controlled release of these agents and extending their antimicrobial efficacy (Fig. 6F) [194].
MXene materials, such as Ti₃C₂Tₓ, also exhibit notable photothermal effects under NIR irradiation, directly damaging or inducing apoptosis in tumor cells [195]. Yin et al. [185] developed a 3D-printed bone-mimicking scaffold (NBGS) incorporating 2D niobium carbide (Nb₂C) MXene nanosheets. This scaffold achieved dual functionality for osteosarcoma therapy and bone defect repair. Nb₂C MXene exhibited excellent photon responsiveness in the second NIR window, efficiently ablating osteosarcoma cells through photothermal effects. Concurrently, the degradation of the scaffold released Nb-based substances, promoting angiogenesis and delivering oxygen and nutrients to support bone repair. Additionally, the release of calcium and phosphate ions during degradation accelerated new bone tissue mineralization. This multifunctional approach, integrating tumor photothermal ablation, angiogenesis promotion and osteogenesis, highlighted immense potential of MXenes in osteosarcoma treatment and BTE. Building on this, Yang et al. [196] introduced a mesoporous structure into similar NBGS to enable precise and controlled nitric oxide (NO) release. Gradual release of low-concentration NO significantly promoted angiogenesis, demonstrating an intelligent and adjustable biomaterial platform.
In conclusion, MXenes represent an emerging class of multifunctional 2D nanomaterials that offer remarkable advantages for orthopedic applications, including high electrical conductivity, photothermal responsiveness, antimicrobial activity and osteoinductive potential. Their ability to modulate bone regeneration, eradicate pathogens and facilitate tumor ablation positions them as promising candidates for the next generation of smart bone implants and therapeutic scaffolds.
(A) The structure of the MAX phase and the corresponding MXenes obtained by chemically selectively etching the A layer. Reprinted with permission from Ref [184]. Copyright 2014, John Wiley and Sons. (B) The integration of Nb2C into BG can be used for photothermal-driven osteosarcoma ablation therapy with potent angiogenesis and osteogenic effects. Reprinted with permission from Ref [185]. Copyright 2021, Yin et al. (C) Diagrammatic illustration of a ceramic bracket decorated with Ti3C2Tx. Promotes bone healing by channeling dynamic inflammation and bone tissue responses. Reprinted with permission from Ref [186]. Copyright 2024, Huang et al. (D) Schematic Nb2C@TP of multimodal infection control biofunctions with bacterial clearance and tissue regeneration capabilities. Reprinted with permission from Ref [188]. Copyright 2021, American Chemical Society. (E) MXene loaded electroactive hydrogels promote efficient bone regeneration in an electro-microenvironment. Reprinted with permission from Ref [192]. Copyright 2022, Hu et al. (F) Schematic diagram of the synthesis of MXene-based antimicrobial nanosystems, presenting promising antimicrobial and anti-inflammatory strategies. Reprinted with permission from Ref [194]. Copyright 2023, John Wiley and Sons. Nb2C, niobium carbide; TP, titanium plate
Others
In the exploration of advanced materials for orthopedic applications, emerging candidates such as metal-organic frameworks (MOFs), hBN and g-C3N4 offer immense potential despite their relatively nascent stage of application in this field. These materials demonstrate unique physicochemical properties, including exceptional tunability, mechanical resilience and bioactivity, making them promising for addressing complex challenges in bone repair, osteogenesis and antibacterial therapy. Nonetheless, current research predominantly focuses on their fundamental properties and preliminary biomedical explorations, highlighting the need for comprehensive preclinical studies to fully unlock their translational potential in clinical settings.
MOFs
MOFs, composed of metal ions or clusters coordinated with organic ligands, exhibit highly tunable porosity, extensive specific surface areas and remarkable chemical and thermal stability. These porous structures enable efficient loading of a wide range of osteogenic, anti-tumor drugs as well as bioactive molecules, such as plasmids, non-coding RNAs, exosomes or autophagy activators, making them promising candidates for treating diseases like OA, bone defects, avascular necrosis and osteosarcoma [197,198,199,200,201,202]. Two-dimensional metal–organic frameworks (2D MOFs) possess a higher specific surface area, and their ultrathin thickness maximizes exposure of surface-active metal sites, making them highly amenable to modification. Consequently, they are increasingly favored in orthopaedic applications. Qiao et al. developed a medium-pressure response system based on an interface-limited 2D MOF grown on carbon nanotubes (CNTs). The ultra-small copper-based 2D MOF exhibited abundant surface and interface defects, endowing the system with numerous surface states (Fig. 7B). Under microwave (MV) irradiation, the CNT-2D MOF effectively absorbed microwaves and converted them into heat via enhanced heterointerface polarization, enabling microwave thermal therapy. Simultaneously, surface states facilitated generation of excited electrons for microwave dynamic therapy. This CNT-2D MOF system demonstrated highly efficient broad-spectrum antibacterial activity against seven pathogenic bacteria, including both Gram-negative and Gram-positive strains, after only 7 min of MV irradiation. Cell viability and morphology assays following co-cultivation with CNT-2D MOF revealed no significant differences compared to controls. Although increasing concentration slightly reduced cell viability, even at 1 mg/mL viability remained above 94%. Hemolysis assays with rabbit red blood cells showed no significant hemolysis, with only a 6.3% hemolysis rate observed at 1 mg/mL. Furthermore, liver and kidney function indicators in vivo showed no significant differences between CNT-2D MOF-treated groups and controls, suggesting favorable in vivo safety and efficient clearance. In a rabbit osteomyelitis model, microwave treatment raised local temperature from 28 °C to 42 °C within 7 min, demonstrating good in vivo thermal stability. After 5 days of treatment, Staphylococcus aureus counts in bone marrow decreased dramatically to 0.619 × 10 CFU/g compared to 7.181 × 10 CFU/g in controls, evidencing potent antibacterial efficacy [203].
Given the complex pathophysiological environment in orthopaedic diseases, the performance, in vivo stability, and degradation kinetics of 2D MOFs are critical. For instance, the local inflammatory milieu in arthritis may affect MOF stability and function. Shi et al. synthesized 2D manganese porphyrin-based MOF nanosheets (ZMTP) via coordination of MnTCPP + with Zn²⁺ benzoate oxygen groups, using them as antioxidant nanoenzymes mimicking superoxide dismutase (SOD) and catalase activities (Fig. 7A). Step potential shift measurements in electrolytes indicated high redox stability. Long-term degradation assessed by dispersing ZMTP in weakly acidic PBS revealed a two-stage degradation: initial small pore formation after 12 h, progressive pore enlargement over 24 h, and near-complete degradation by 96 h, indicating initial maintenance of catalytic activity followed by degradation and clearance ensuring biosafety. In an arthritis mouse model, ZMTP treatment significantly downregulated pro-inflammatory cytokines in joints and upregulated alkaline phosphatase (ALP) levels, demonstrating effective anti-arthritis and enhanced osteogenic outcomes. Histological analyses of major organs (heart, liver, spleen, kidneys) showed negligible pathological changes, confirming good in vivo clearance [204].
Bone defects following tumour resection often resist self-repair and risk tumour recurrence or infection, necessitating antibacterial bone fillers with robust in vivo stability. Certain 2D MOFs excel due to their ultrathin layers, high surface area, and extensive π-conjugated rings, which enable rapid response to external light, high photothermal conversion efficiency, and excellent stability. Wu et al. fabricated 2D cobalt-coordinated tetra(4-carboxyphenyl)porphyrin (Co-TCPP) MOF nanosheets exhibiting ultrathin thickness and large surface area, facilitating swift light responsiveness and efficient photothermal conversion. The π-conjugated porphyrin macrocycles confer remarkable photothermal stability. Incorporated into calcium phosphate cement (CPC), 2D Co-TCPP MOF accelerated CPC degradation, likely due to the relatively high dissolution rate of Co-TCPP, without compromising photothermal performance. Under laser irradiation, the 1% Co-TCPP/CPC composite rapidly increased temperature, maintaining above 50 °C, resulting in significant tumour necrosis reduction. The injectable paste conformed well to bone defect morphology. Over an 8-week period, CPC degraded at the defect site while promoting extensive new bone formation and neoangiogenesis. These results indicate that 2D MOF nanosheets maintain the intrinsic properties of CPC, with stable degradation and photothermal performance conducive to orthopaedic tumour treatment [205].
h-BN
hBN, often referred to as white graphene, is a 2D crystalline nanomaterial composed of boron and nitrogen atoms arranged in a hexagonal honeycomb lattice and its physicochemical properties as well as structural characteristics bear a striking resemblance to those of graphite crystals [206]. The remarkable combination of high tensile strength and exceptional chemical inertness has facilitated its extensive exploration within the medical domain. At present, hBN nanosheets are predominantly being engineered for advanced bioimaging applications and drug delivery systems, particularly in oncological therapeutics.
Despite its immense potential, research into the utilization of hBN for orthopedic disorders remains relatively nascent. Experimental evidence has elucidated that the incorporation of hBN could markedly augment the mechanical robustness of composite materials, thereby enabling the fabrication of superior bone-regenerative scaffolds [207, 208]. For instance, Ensoylu et al. [209] successfully integrated hBN nanoparticles (0.1%−2%) into borate-based BG scaffolds, observing a pronounced enhancement in compressive strength and in vitro mineralization capabilities. Concurrently, drug release studies revealed that hBN expedited the release kinetics of gentamicin and 5-fluorouracil, adhering to first-order and Higuchi models respectively. In addition, the antibacterial attributes of hBN render it exceptionally promising for mitigating bone infection risks. Mukheem et al. [210] embedded hBN within a polyhydroxyalkanoate/chitosan (PHA/Ch) polymeric matrix leveraging its thermal stability and nanoscale morphology. This sophisticated nanocomposite exhibited potent bactericidal efficacy against MRSA and multidrug-resistant E. coli K1 strains, while maintaining excellent cytocompatibility with negligible cytotoxicity. These findings underscore its viability as a foundational element in the development of antibacterial scaffolds for biomedical applications.
Research by Goncu et al. illuminated the profound impact of hBN on the rheological behavior of hydrogel matrices. The intrinsic lubricity of hBN allowed its uniform dispersion within the hydrogel network, significantly diminishing zero-shear viscosity and deformation resistance. Such rheological refinements highlight that hBN enhances the dynamic synergy of hydrogels, offering compelling theoretical underpinnings for its prospective deployment in joint lubrication and OA therapeutics (Fig. 7E) [211].
The application of hexagonal boron nitride (hBN) in orthopaedics remains at an early stage, with several challenges requiring attention to achieve consistent dispersion within composite scaffolds. The intrinsic chemical inertness and hydrophobicity of hBN nanoparticles hinder their stable dispersion in aqueous or biocompatible solvents. Moreover, strong van der Waals interactions among hBN nanoparticles often lead to agglomeration, resulting in uneven distribution within the scaffold and localized stress concentration. Manufacturing processes further influence hBN dispersion; for instance, shear forces during 3D printing can promote nanoparticle aggregation, while ice crystal formation during freeze-drying may compress hBN particles, causing localized enrichment zones. Therefore, optimizing dispersion techniques is imperative, including surface functionalisation approaches or the development of effective and stable dispersants and stabilizers. Composite dispersion strategies, such as combining mechanical mixing with ultrasonic treatment, can also be employed to enhance uniformity during scaffold fabrication.
Regarding biocompatibility, Bianco et al. reported that hBN exerts lower toxicity on dendritic cell (DC) activity and DC-mediated T cell polarization compared to MoS₂ and graphene oxide (GO) [212]. Some studies combining hBN with osteoblast-like cells observed decreased cell viability, potentially due to nanoparticle internalization triggering reactive oxygen species (ROS) generation. Conversely, other research found no cytotoxic effects at hBN concentrations below 22 µg/mL, with indications of reduced oxidative stress. At low concentrations, hBN does not induce mitochondrial depolarization or disrupt F-actin cytoskeleton formation [213]. Nevertheless, investigations into the cellular compatibility of hBN remain limited. Crucially, the long-term effects of boron released from hBN, including local bone tissue accumulation and systemic distribution, as well as safety thresholds, require further elucidation. The biodegradation mechanisms and metabolic pathways of hBN are yet to be fully understood. Inefficient clearance of nanoparticles may provoke chronic inflammatory responses and organ damage. Furthermore, current studies predominantly focus on in vitro assays or short-term in vivo experiments. Essential data on the long-term in vivo behavior of hBN—such as biocompatibility, particle release kinetics, chronic inflammation, and organ accumulation—remain insufficient and warrant comprehensive investigation.
g-C3N4
g-C3N4 is an emerging visible-light-responsive non-metallic semiconductor material. Davardoostmanesh et al. [214] demonstrated that g-C3N4 nanosheets, prepared via electrophoretic size fractionation, exhibit exceptional photoluminescence properties. Compared to bulk g-C3N4, these nanosheets display significantly higher cytotoxicity against human osteosarcoma cell line Saos-2, while exhibiting negligible toxicity to normal human foreskin fibroblasts. Similarly, Taheri et al. [215] reported that photoactivated g-C3N4 can effectively induce cancer cell death without the need for additional photosensitizers or chemotherapeutic agents, as it is efficiently internalized by tumor cells in vitro. The underlying mechanism involves the activation of pathways associated with oxidative stress, cell death and apoptosis, with its anticancer effect being strictly light-dependent. For normal cells, g-C3N4 shows negligible cytotoxicity. This is attributed to the selective accumulation of photosensitizers in tumour tissue combined with precise spatial control of light exposure, which ensures that ROS generation occurs mainly within tumour cells or their microenvironment. Given their extremely short lifespan and limited action range, ROS cytotoxicity is strictly confined to areas with concentrated photosensitizers under light exposure. Moreover, tumour cells exist in a state of oxidative stress, rendering them more susceptible to ROS-induced damage, thereby enhancing the selective cytotoxicity towards tumour cells. However, because ROS lack intrinsic specificity, misdirected delivery of g-C3N4 to healthy tissues or improper control of light exposure may result in damage to healthy bone tissue. Therefore, further research is necessary to enhance targeted delivery of photosensitizers and achieve precise control of light exposure, thereby minimizing ROS-induced damage to healthy tissues.
In the context of osteogenesis, g-C3N4 has also demonstrated remarkable potential. Sadek et al. [216] revealed that g-C3N4 upregulates osteogenic marker genes such as Col I, OCN and osteopontin in vitro and significantly enhances bone healing in a rabbit critical-sized femoral condyle defect model. This is evidenced by higher percentages of bone-like tissue, increased collagen maturation and improved biodegradability. Additionally, Dutta et al. [217] found that oxidized g-C3N4 (Ox-gCN), when incorporated into alginate/gelatin hydrogel scaffolds, significantly boosts the adhesion, proliferation and osteogenic differentiation of hBMSCs. This is achieved through the enrichment of oxygen-containing functional groups on its surface and the activation of ERK and MAPK signaling pathways, which further promote macrophage M2 polarization and the expression of anti-inflammatory genes, thereby facilitating bone immunomodulation. Moreover, its visible-light-induced photocatalytic properties efficiently generate ROS, inhibiting bacterial biofilm formation and preventing infection. In a rat calvarial defect model, the Ox-gCN scaffold expressed superior hemostatic, anti-inflammatory and bone regeneration capabilities, establishing itself as a multifunctional platform for the repair of traumatic bone injuries (Fig. 7F).
Despite these advancements, the clinical translation of these materials still faces significant challenges. Although biodegradable materials have been explored in experimental settings, their in vivo safety and long-term effects remain insufficiently investigated. Moreover, the degradation kinetics of these materials in animal models are inadequately characterized, resulting in critical knowledge gaps. Additionally, regulatory pathways and standardized evaluation systems for these materials are underdeveloped, contributing to prolonged timelines for clinical translation.
From a manufacturing perspective, production often involves complex solvothermal conditions and incurs high costs. Scale-up processes face challenges such as poor batch-to-batch consistency and elevated purification expenses. Even slight variations in manufacturing parameters can substantially affect material performance. Furthermore, the behavior of these materials in the complex pathophysiological environment of the human body must be carefully considered. Patient-specific factors, including age, metabolic diseases, and genetic background, may influence material-host interactions, thereby affecting the reproducibility and efficacy of clinical applications. Overcoming these translational bottlenecks necessitates multidisciplinary collaboration. Future research should focus on strategies such as surface functionalization to enhance biocompatibility and biodegradability, alongside rigorous and comprehensive toxicity evaluations—particularly within immunocompromised and bone defect models—to support safer and more effective clinical translation.
There is also a need for modular integration of these materials into composite platforms—such as 3D-printed scaffolds, injectable systems, and responsive coatings—to improve their clinical usability. Interdisciplinary collaboration will be crucial to unlock the full potential of these materials, linking advances in nanomaterial science with orthopedic surgery, microbiology, and tissue engineering. With further mechanistic elucidation and translational design, these emerging materials are well positioned to supplement or even transcend conventional bone substitutes, particularly in complex cases involving infection, inflammation, or tumor recurrence.
(A) Schematic diagram of the mechanism of action of ZMTP nanosheets in the treatment of rheumatoid arthritis using nanocatalysis. Mimics the activity of superoxide dismutase (SOD) and catalase, downregulates oxidative stress, and promotes osteogenesis. Reproduced with permission from Ref [204]. Copyright 2022, Springer Nature. (B) Schematic diagram of the microwave absorption mechanism of CNT-CuHHTP. Exhibits antibacterial therapeutic effects in osteomyelitis. Reprinted with permission from Ref [203]. Copyright 2023, John Wiley and Sons. (C) hBN added to hydrogels can reduce the dynamic viscosity and deformation rate of polymers with zero shear points and is expected to be a drug candidate for the treatment of osteoarthritis. Reprinted with permission from Ref [211]. Copyright 2023, American Chemical Society. (D) Ox-gCN-modified hydrogel scaffolds achieve anti-inflammatory and bone immunomodulation for traumatic bone injury repair. Reproduced with permission from Ref [217]. Copyright 2024, Elsevier. MOF, metal-organic framework; β-TCP, beta-tricalcium phosphate; ROS, reactive oxygen species; Ox-gCN, oxidized g-C3N4

